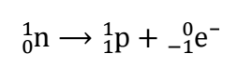What is radiation?
|
NEW VIDEO
|
|
|
Discovery of Radioactivity
In 1895, while conducting experiments using a cathode ray tube, German physicist Wilhelm Roentgen discovered invisible rays that could cause a distant viewing screen to glow even when blocked by cloth, paper, or books. Roentgen labeled these rays “X” due to their unknown nature, leading to the eventual name X-rays. In 1901, Roentgen was awarded the first Nobel Prize in Physics for his discovery.
In 1895, while conducting experiments using a cathode ray tube, German physicist Wilhelm Roentgen discovered invisible rays that could cause a distant viewing screen to glow even when blocked by cloth, paper, or books. Roentgen labeled these rays “X” due to their unknown nature, leading to the eventual name X-rays. In 1901, Roentgen was awarded the first Nobel Prize in Physics for his discovery.
Within months of Roentgen’s discovery of X-rays, French physicist Henri Becquerel discovered that crystals of uranium salt emitted a similar form of radiation without any external stimulation. Becquerel had made the first observation of spontaneous radiation, which later became known as radioactivity, and isotopes that spontaneously transform into other nuclei are called radioactive. The search immediately began for other naturally radioactive materials.
Marie Curie, a naturalized French physicist, was among the first to recognize the significance of Becquerel’s discovery. She began measuring the radioactivity of other minerals containing uranium, including pitchblende and torbernite. Notably, Curie discovered that the amount of radioactivity depended only on the amount of uranium present in a mineral, and correctly deduced that the radiation must result from changes in uranium’s atomic structure. Curie’s husband Pierre eventually abandoned his own research on crystals to assist his wife in the laboratory (Figure 5).
The Curies began the painstaking work of separating the radioactive elements from minerals and documenting their level of radioactivity. In July 1898, they announced the discovery of a new radioactive element named “polonium” after Marie’s original homeland of Poland. Later that year they successfully isolated a second undiscovered element, radium. Marie and Pierre Curie, along with Henri Becquerel, were jointly awarded the 1903 Nobel Prize in Physics for their discoveries regarding the nature of radioactivity. Marie Curie also won the 1911 Nobel Prize in Chemistry, making her the only person in history to be awarded two Nobel Prizes in scientific categories.
The dangers of radiation exposure were not understood in the decades surrounding its initial discovery. By the 1930s, Marie Curie’s near-constant exposure to high levels of radiation had slowly deteriorated her health and severely weakened her eyesight. Curie died in 1934 from aplastic anemia, believed to be a direct result of radiation exposure. Curie’s original research documents from the 1890s have been so contaminated by radiation that they must be kept constantly shielded even today.
In 1900, Ernest Rutherford and Frederick Soddy began documenting the forms of radiation emitted by radioactive materials. They were the first to conclude that radioactivity is due to unstable elements spontaneously transforming into different elements. This process of transformation is generally referred to as radioactive decay. Rutherford identified three different “rays” which he named alpha (α), beta (β), and gamma (γ) after the first three letters of the Greek alphabet.
The three types of radiation exhibited different properties. For instance, each ray was deflected in a different direction by magnetic fields, indicating that they had different electric charges. Alpha rays are positively charged, beta rays are negatively charged, and gamma rays have no electric charge. Subsequent analysis eventually revealed that each form of radiation was a known particle: alpha rays are helium nuclei, beta rays are electrons, and gamma rays are high-energy photons. (Although we now recognize them as particles, the terms “alpha ray” and “beta ray” are occasionally still used as a matter of historical convention.)
Each ray also has different penetration strengths in matter. Due to its larger mass, an alpha particle penetrates the least into ordinary matter and can be blocked with an ordinary sheet of paper. Beta particles, being less massive, will pass through paper but can be stopped by a sheet of metal such as aluminum. Gamma rays are the most penetrating form of radiation because they consist of high-energy photons with no mass. Gamma radiation can only be stopped by several centimeters of lead shielding.
We will discuss each type of decay in further detail.
We will discuss each type of decay in further detail.
ALPHA-BETA-GAMMA Detector
|
|
|
Alpha Decay
An atom undergoes alpha decay by releasing an alpha particle from its nucleus. An alpha particle consists of two protons and two neutrons bound together. After releasing an alpha particle, the nucleus’ neutron number N and atomic number Z have both decreased by 2, and its mass number A has decreased by 4. This is represented with a reaction “equation” analogous to chemical reactions:
An atom undergoes alpha decay by releasing an alpha particle from its nucleus. An alpha particle consists of two protons and two neutrons bound together. After releasing an alpha particle, the nucleus’ neutron number N and atomic number Z have both decreased by 2, and its mass number A has decreased by 4. This is represented with a reaction “equation” analogous to chemical reactions:
In this notation, X is called the parent nucleus and Y is called the daughter nucleus. For example, a well-known example of alpha decay is the decay of uranium-238 to produce thorium-234, which can be represented as
Equations that describe radioactive decay must follow several “rules” in order to be balanced. First, the sum of the atomic numbers on the left side of the arrow must equal the sum of the atomic numbers on the right side of the arrow. Second, the nucleon number must stay the same, meaning the sum of the mass numbers on the left must equal the sum on the right.
Alpha decay is most common among heavier atoms, with tellurium (Z = 52) being among the lightest elements known to undergo alpha decay. Alpha particles are often emitted at tremendously high speeds, around 5% the speed of light. However, due to their relatively large mass, they often tend to give up their energy of motion due to collisions within a few centimeters from their source.
Beta Decay
During beta decay, the mass number A remains constant, but Z either increases or decreases by 1 in order to conserve charge:
For example, carbon-14 decays into nitrogen-14 through β- decay:
Note that the emitted electron originates from the nucleus and not from the atom’s electron cloud. How is it possible for the nucleus to emit an electron when there are no electrons within the nucleus? As it turns out, the electron is created in the nucleus during beta decay when a neutron transforms into a proton. We can represent this process as:
Even outside the nucleus, free neutrons are unstable and will undergo beta decay with an average lifetime of around 15 minutes.
Studies of beta decay during the early 20th century revealed some unusual behavior. For instance, the beta particles emitted during beta decay almost always had less energy than the laws of physics predicted. Furthermore, other quantities such as angular momentum did not appear to be conserved during beta decay, which led many prominent physicists such as Niels Bohr to question the existing laws of physics.
In 1930, Wolfgang Pauli hypothesized that an additional particle must be emitted during beta decay in order to account for the missing energy and momentum. Enrico Fermi named this particle neutrino, or “little neutral one,” in order to distinguish it from the neutron. The neutrino, emitted during β+ decay, is represented by the symbol ν (a Greek lowercase “nu”), whereas the antineutrino, emitted during β- decay, is represented by ν̅. (The antineutrino is the antiparticle of the neutrino.) The neutrino has zero electric charge and an extremely small mass. Due to its weak interaction with matter, the neutrino was not experimentally observed until 1950.
Gamma Decay
Gamma decay is the process by which a nucleus transitions from an excited energy state to a lower energy state, releasing energy in the form of a gamma ray. Unlike alpha and beta rays, gamma rays are high-frequency, high-energy electromagnetic waves. Gamma decay does not change the atomic number or atomic mass number of a nucleus. It is common to use an asterisk (*) to denote a nucleus in an excited energy state, as in this example:
Gamma decay is the process by which a nucleus transitions from an excited energy state to a lower energy state, releasing energy in the form of a gamma ray. Unlike alpha and beta rays, gamma rays are high-frequency, high-energy electromagnetic waves. Gamma decay does not change the atomic number or atomic mass number of a nucleus. It is common to use an asterisk (*) to denote a nucleus in an excited energy state, as in this example:
It is possible for nuclei to become excited following a high-energy collision with another particle. More commonly, however, nuclei are left in an excited state immediately after undergoing alpha or beta decay.