Radioactive Decay & Half-life
Radioactive decay is a spontaneous event that takes place randomly, and there is no way to predict when any individual radioactive nucleus will undergo decay. However, we can use probabilities to describe how many nuclei within a given sample can be expected to undergo decay within a certain time period.
It is common to describe the decay rate of a radioactive isotope by its half-life. The half-life of a radioactive isotope is defined to be the time it takes for, on average, half of a given sample of nuclei to decay. After one half-life, half of the original nuclei will remain. After two half-lives, one quarter will remain. After three half-lives, one eighth will remain, and so on (Figure 6).
|
|
|
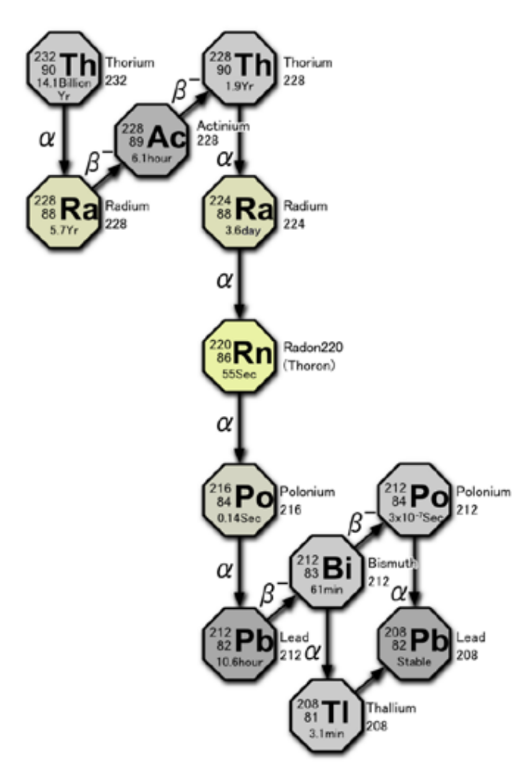
Decay Chains
Not all nuclei decay into stable nuclei. In some cases, a parent nuclei decays into a radioactive daughter nucleus, which then becomes the parent for a subsequent decay process. This phenomenon is referred to as a decay chain or decay series. Note that subsequent decays do not happen immediately; each decay process along the chain will have its own characteristic half-life, which may differ greatly from the others.
Not all nuclei decay into stable nuclei. In some cases, a parent nuclei decays into a radioactive daughter nucleus, which then becomes the parent for a subsequent decay process. This phenomenon is referred to as a decay chain or decay series. Note that subsequent decays do not happen immediately; each decay process along the chain will have its own characteristic half-life, which may differ greatly from the others.
Detection of Radioactivity
There are several different methods of detecting the presence of radiation, most of which rely on its ability to ionize (strip electrons from) ordinary atoms and molecules.
There are several different methods of detecting the presence of radiation, most of which rely on its ability to ionize (strip electrons from) ordinary atoms and molecules.
A Geiger counter consists of a low-pressure tube of inert gas surrounding a positively charged wire. In the presence of radiation, high-energy particles collide with atoms in the gas and knock electrons loose. These freed electrons are attracted to the wire, and collide with other atoms to free other electrons along the way. The result is a brief electric pulse that triggers a counter attached to the tube, often accompanied by the familiar “click” sound associated with the device. Geiger counters are usually compact enough to be used as a hand-held device. Hans Geiger, one of the researchers in Rutherford’s gold foil experiment, developed the principle behind the Geiger counter.
Radiation detectors are also capable of tracing the path of ionizing radiation. A cloud chamber is a sealed chamber containing a vapor of water or alcohol. Radiation passing through the chamber leaves a trail of ions behind it. If the saturated vapor within the chamber is suddenly cooled via a moveable piston, it will condense around these ions, forming cloud-like trails that make the path of the radiation visible. The positron and the muon, another electron-like particle, were both discovered using a cloud chamber detector.
The bubble chamber, invented by Donald Glaser in 1952, is another type of detector that contains liquid hydrogen heated under pressure to just below boiling. When a particle enters the detector, the pressure in the chamber is suddenly decreased, and a trail of microscopic bubbles forms behind the particle. Cameras mounted around the chamber capture the particle trail before it dissipates (Figure 9). When a strong electric or magnetic field is applied, charged particles will follow curved paths. The direction and curvature of these paths provide information about the charge, mass, and momentum of the particles, which aids in their identification. Positive and negative charges, for instance, will be deflected in opposite directions in a magnetic field.